A terminal brain cancer patient, who had relapsed after 17 CAR-T treatments, saw his tumor completely disappear within 4 weeks of receiving a novel TIL therapy and has remained tumor-free for over 20 months.
For inquiries about new cancer therapies and clinical trials:
Phone: 4008803716 | WeChat: 17801183037 | Email :100085_010@163.com
A 56-year-old male patient was diagnosed with left frontal lobe glioblastoma (IDH wild-type, WHO Grade 4, EGFR amplification)—one of the most malignant and difficult-to-treat types of brain glioma.
After undergoing tumor resection on January 5, 2023, he initially received standard treatment, but his condition relapsed after a brief period of stability. Subsequently, he underwent a cumulative total of 17 CAR-T treatments targeting two different antigens, yet his disease continued to progress.
In desperation, he enrolled in a clinical trial for a novel TIL therapy. The results were astounding: just 4 weeks after the TIL cell infusion, imaging examinations showed the tumor in his brain had been completely eliminated, achieving a complete response, and he has remained tumor-free for over 20 months.
—
Fighting for Survival in a Desperate Situation
Glioblastoma is known as the “number one intracranial killer.” The moment of diagnosis feels like the start of a life countdown.
After Mr. W’s diagnosis, he first underwent a high-risk brain tumor resection, followed by temozolomide chemotherapy and radiotherapy. However, his condition was not controlled.
Subsequently, he tried two different CAR-T therapies targeting different antigens, receiving more than 10 intratumoral injections cumulatively, yet this still failed to halt tumor progression.
Traditional treatments have limited efficacy against advanced brain glioma. Even with combined treatment plans involving surgery, radiotherapy, and chemotherapy, the median overall survival for patients is only about 14.6 months.
Facing such a hopeless situation, Mr. W and his family felt profound despair until they learned about the TIL therapy clinical trial.
TIL Therapy
TIL therapy, or Tumor-Infiltrating Lymphocyte therapy, is an innovative cellular immunotherapy.
This therapy starts with the patient’s fresh tumor tissue as the raw material. After isolation, modification, and expansion in vitro, the cells are reinfused into the patient, achieving precise treatment that “fights the tumor with itself.”
Unlike traditional CAR-T therapy, the GC101 TIL therapy does not require lymphodepletion nor IL-2 injections, significantly improving treatment safety and convenience.
Developed by Juncell Therapeutics, GC101 TIL therapy is the world’s first natural TIL therapy that requires no lymphodepletion or IL-2 injection, leveraging its self-developed DeepTIL® cell expansion platform.
Existing clinical data show that GC101 has an objective response rate exceeding 35% against various types of advanced solid tumors. Several patients have achieved complete tumor elimination and complete response, with the longest tumor-free survival duration exceeding 3 years.
The Treatment Process
In late 2023, Mr. W successfully enrolled in the GC101 TIL therapy clinical trial.
The medical team used TIL cells cultured from his surgical sample obtained a year earlier, which had been cryopreserved until needed.
The treatment team successfully expanded the cells to a total of 2.95 × 10^10 TIL cells, with T-cells accounting for a high 99.92% of the population, predominantly CD8⁺ T-cells (97.85%).
After preconditioning, Mr. W received the TIL cell infusion on December 12, 2023.
During the infusion, Mr. W experienced no severe reactions, only a mild fever that subsided quickly. Compared to traditional TIL therapies, this modified version demonstrated higher safety.
A Dramatic Reversal
Just 4 weeks after the TIL cell infusion, imaging examinations on January 8, 2024, revealed that the tumor in Mr. W’s brain had been completely eliminated.
Even more remarkably, as of October 2025, the patient has maintained a tumor-free survival for over 1.8 years (calculated at the time of the news release, this is over 20 months), regaining a normal life.
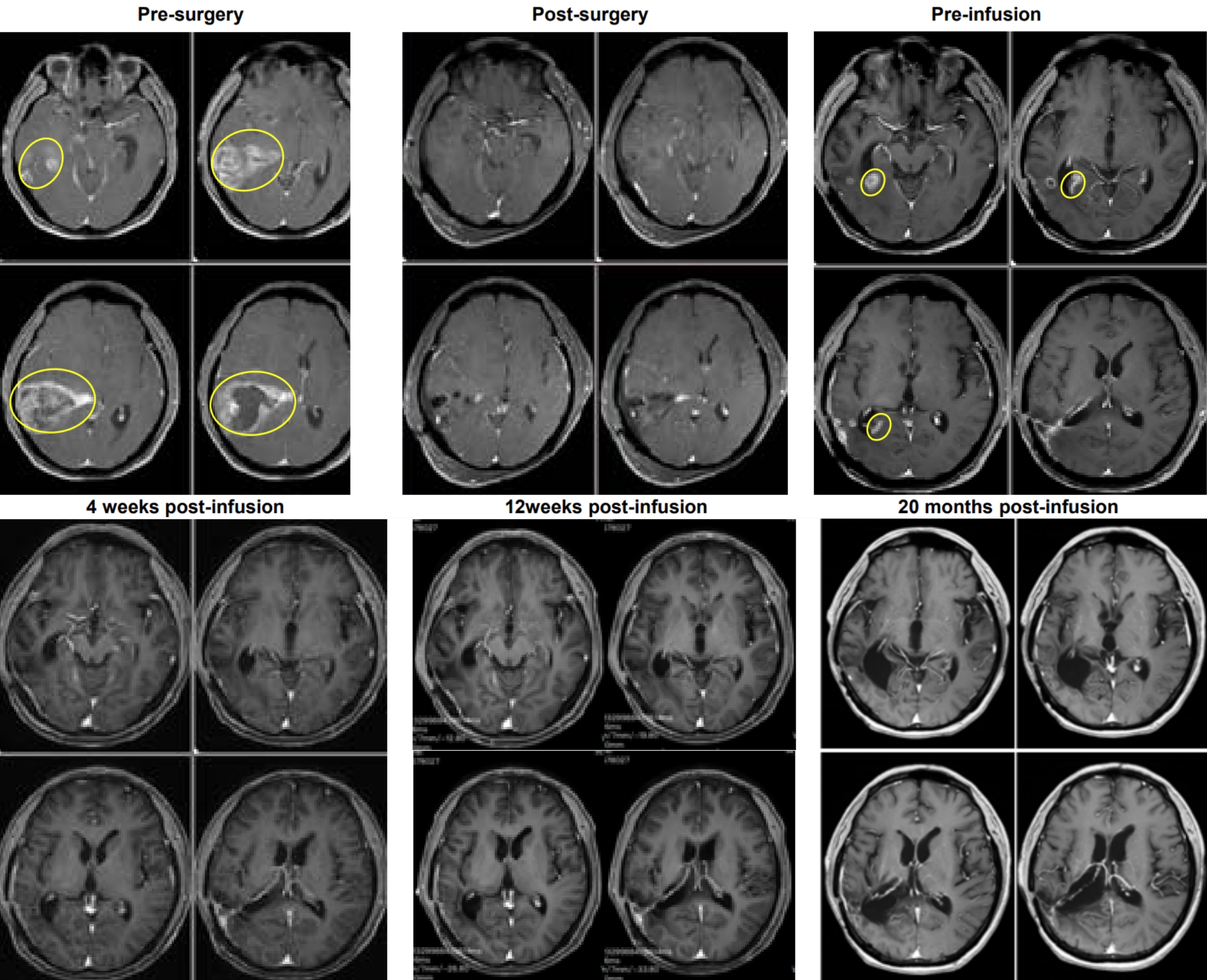
This case sets a new survival record for recurrent glioblastoma and provides crucial clinical evidence for the treatment strategy of “early tissue sampling, TIL reinfusion upon recurrence.”
This remarkable achievement will be presented in a poster at the 40th Annual Meeting of the Society for Immunotherapy of Cancer , taking place from November 7-9, 2025, showcasing it to global industry experts.
Future Prospects
TIL therapy has shown potential not only in brain glioma treatment but also demonstrated remarkable efficacy against other solid tumors like advanced melanoma and non-small cell lung cancer.
In February 2024, the world’s first TIL cell therapy, Lifileucel, received FDA approval in the United States for treating patients with advanced melanoma who had previously received PD-1 inhibitor therapy.
Simultaneously, China is actively advancing clinical research on TIL therapy. The key Phase II registrational clinical trial for Juncell Therapeutics’s GC101 TIL injection targeting advanced melanoma has been approved by the Center for Drug Evaluation of the National Medical Products Administration.
Furthermore, on October 14, 2024, China’s first TIL cell drug registrational trial specifically for lung cancer was officially initiated, intended for treating advanced non-small cell lung cancer.
—
At present, there are still many clinical trials for new anti-cancer drugs and technologies in China seeking patients.
Beijing South Region Oncology Hospital International Department provides consultation on new drugs and technologies.
Phone Number: 4008803716
WeChat ID: 17801183037
Email ①: 100085_010@163.com
Email ②: myimmnet@163.com
Post time: Nov-20-2025

